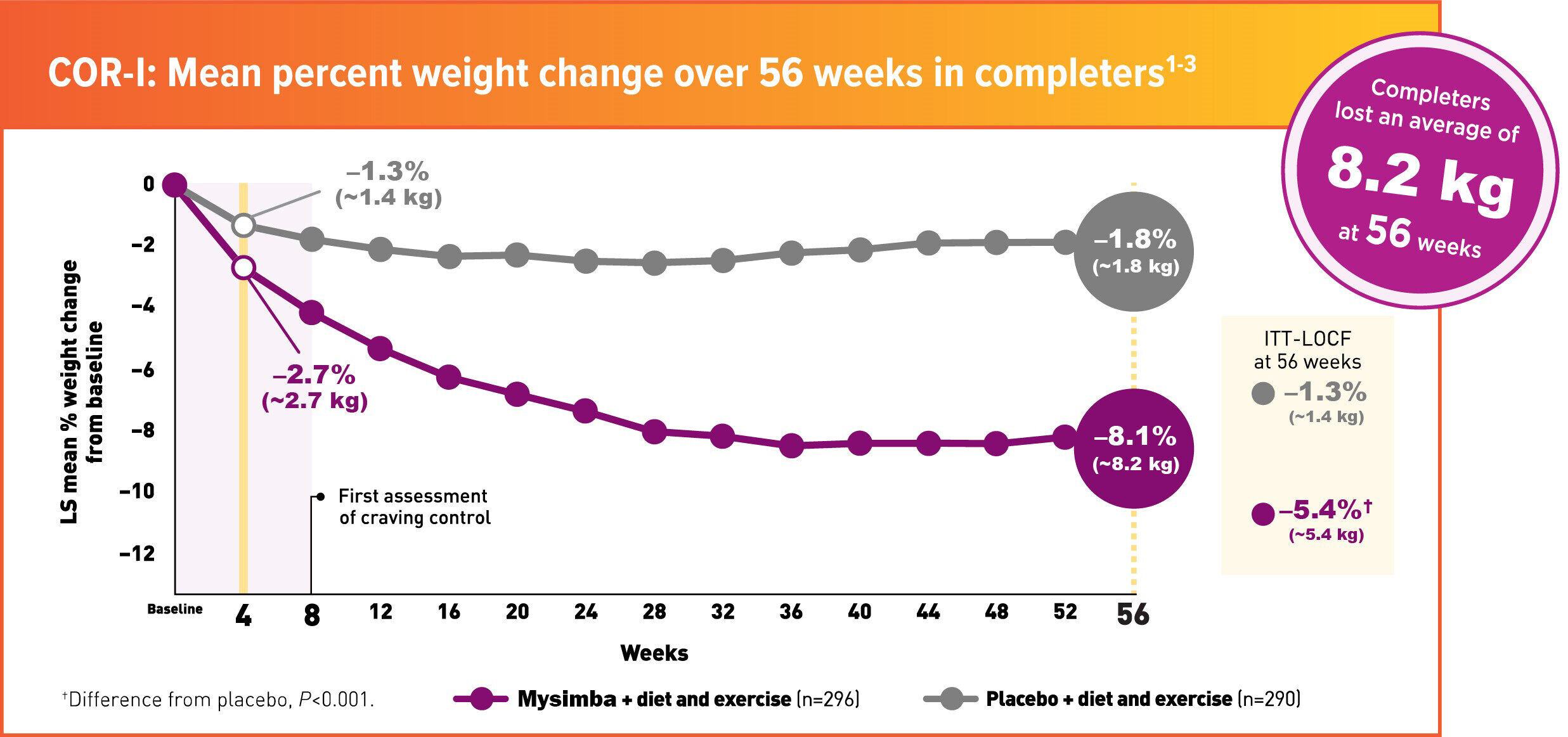
COR-1 Study
Mysimba® Is Proven to Help Patients Lose Weight and Keep It Off
In COR-1, patients were advised to follow a reduced-calorie diet, increase physical activity, and participate in behavioural counseling1
- 62% of patients taking Mysimba® achieved ≥5% weight loss vs 23% taking placebo (completers)1
- 31% of patients taking Mysimba® achieved ≥5% weight loss vs 12% taking placebo (ITT-BOCF)1
ITT=Intention to treat; LOCF= Last observation carried forward; LS=least squares; BOCF; baseline observation carried forward; COR = Contrave Obesity Research
COR-1 was a 56-week, multicentre, double-blind, placebo-controlled study. Patients were randomised to Mysimba® 32 mg/360 mg daily or placebo. The co-primary endpoints were percent change from baseline body weight and the proportion of patients achieving ≥5% reduction in body weight at Week 56. Unless noted otherwise, data shown are from the ITT analysis, which included all randomised patients who had body-weight measurements at baseline and at least once post-baseline. LOCF was used for missing data.1,3
Mysimba® ITT: n=538, placebo ITT: n=536. Average baseline parameters were: Mysimba®: 99.8 kg, 109.2-cm waist circumference; placebo: 99.3 kg, 109.2-cm waist circumference. Patients who completed 56 weeks of treatment: Mysimba®: 50.8%; placebo: 49.9%.1
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu
- Data on file, Currax Pharmaceuticals.
- Greenway FL, Fujioka K, Plodkowski RA, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2010;376(9741):595-605.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Weight loss was primarily due to a reduction in fat mass 1,2
In a sub-study of COR-I, patients taking Mysimba lost significantly more fat mass vs placebo at week 52.
 In an additional analysis, patients taking Mysimba experience significant reduction of visceral fat mass vs placebo at week 52 (secondary endpoint).1,2
In an additional analysis, patients taking Mysimba experience significant reduction of visceral fat mass vs placebo at week 52 (secondary endpoint).1,2
Mysimba (n=34): 0.52 kg decrease from baseline; placebo (n=24): 0.13 kg decrease from baseline.
LS mean difference : 0.37 kg ; P= 0.037

Study design: The COR-I sub-study primary endpoint was change from baseline in body composition, measured by total fat mass using DEXA, in the MYSIMBA and placebo groups at week 52. Eligible patients were selected to undergo body composition analysis using DEXA and visceral fat analysis using multislice CT. The MYSIMBA group combined subjects treated with a lower dose of naltrexone (16 mg/day) and the approved MYSIMBA dose (32 mg/day). Mean total body fat mass at baseline was 42.1 kg in the placebo group and 40.5 kg in the MYSIMBA group. Mean visceral fat mass at baseline was slightly higher for the placebo group (3.8 kg) compared with the MYSIMBA group (4.2 kg). Analysis included all randomized patients who had body-composition analysis at baseline and at least once postbaseline during the defined treatment phase. LOCF was used for missing data. Data presented are LS means.1,2 LS mean change and percent change in total body weight from baseline in this sub-study were -7.0 kg (7.2%) for patients taking MYSIMBA and -2.0 kg (2.1%) for placebo.2 CT=computed tomography; DEXA=dual energy X-ray absorptiometry.
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu ..
- Data on file, Currax Pharmaceuticals.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
COR-Behavioural Modification (BMOD) Study
Mysimba® Enhances Diet and Exercise Regimens for Sustained Long-term Weight Loss
In COR-BMOD, patients participated in 28 group counseling sessions over 56 weeks and received individualized daily caloric goals and a prescribed exercise regimen1,2
- 80% of patients taking Mysimba® achieved ≥5% weight loss vs 60% taking placebo (completers)1
- 46% of patients taking Mysimba® achieved ≥5% weight loss vs 34% taking placebo (ITT-BOCF)1
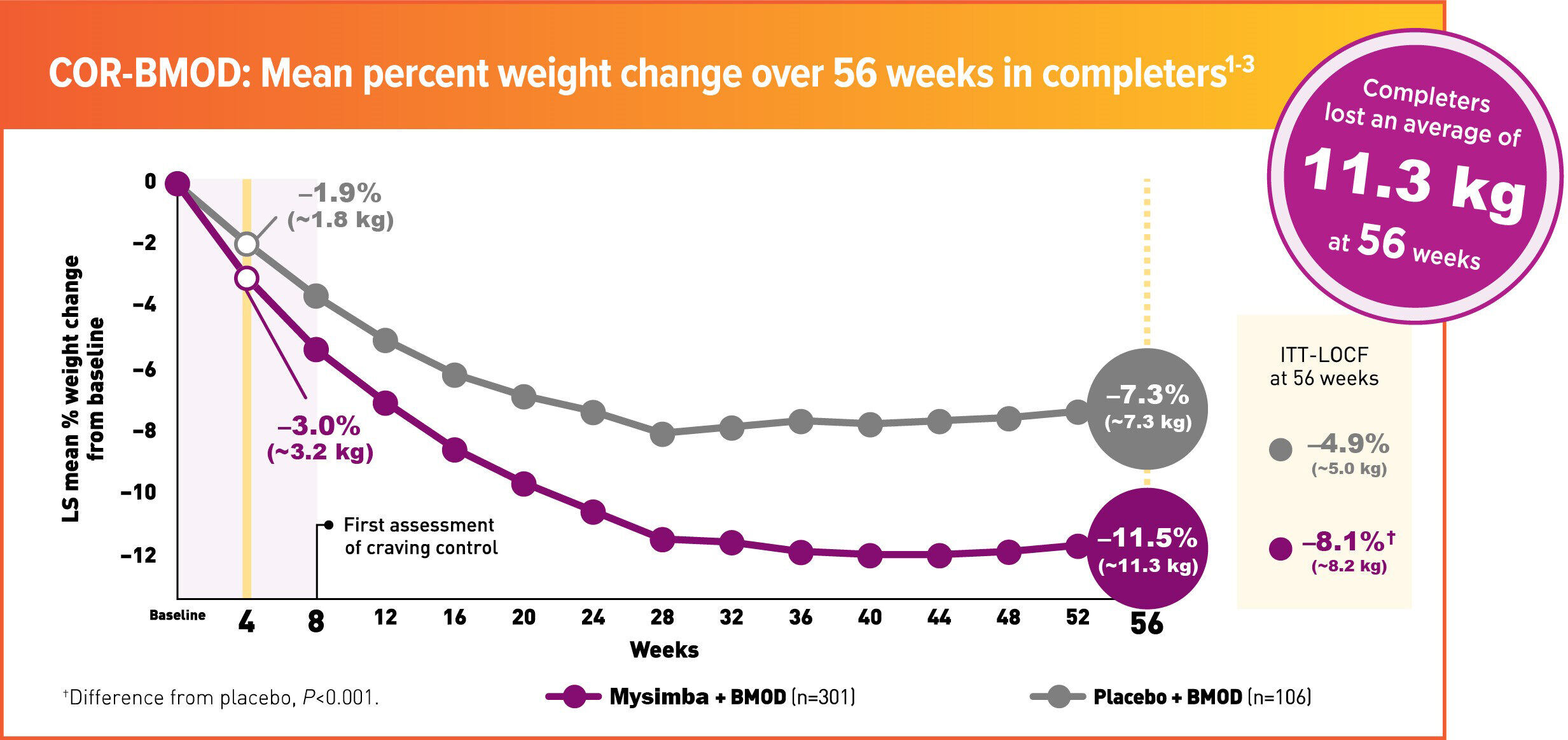
ITT=Intention to treat; LOCF= Last observation carried forward; LS=least squares; BOCF; baseline observation carried forward; COR = Contrave Obesity Research; BMOD = Behavioural Modification.
COR-BMOD was a 56-week, multicentre, double-blind, placebo-controlled study. Patients were randomised to Mysimba® 32 mg/360 mg daily or placebo. The co-primary endpoints were percent change from baseline body weight and the proportion of patients achieving ≥5% reduction in body weight at Week 56. Unless noted otherwise, data shown are from the ITT analysis, which included all randomised patients who had body-weight measurements at baseline and at least once post-baseline. LOCF was used for missing data.1,2
Patients received intensive behavioural modification by registered dietitians, behavioural psychologists, or exercise specialists, as well as a prescribed rigorous diet and exercise regimen. Patients met in groups of 10-20 people for 90 minutes. Group meetings were held weekly for the first 16 weeks, every other week for the next 12 weeks, and monthly thereafter (28 sessions in total).1,2
Mysimba® ITT: n=565, placebo ITT: n=196. Average baseline parameters were: Mysimba®: 100.2 kg, 109.2-cm waist circumference; placebo: 101.6 kg, 109.2-cm waist circumference. Patients who completed 56 weeks of treatment: Mysimba®: 57.9%; placebo: 58.4%.1
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu .
- Wadden TA, Foreyt JP, Foster GD, et al. Weight loss with naltrexone SR/bupropion SR combination therapy as an adjunct to behavior modification: the COR-BMOD trial. Obesity (Silver Spring). 2011(1);19:110-120.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
COR-Diabetes
In COR-Diabetes, A1c reductions were seen when patients with type 2 diabetes lost weight with Mysimba® (mITT-LOCF).1
Mysimba® significantly improved A1c in patients with type 2 diabetes1
- Significantly more patients achieved ≥5% weight loss with Mysimba® (28%) vs placebo (14%) at 56 weeks (P<0.001; ITT-BOCF analysis)1
- Patients lost significantly more weight with Mysimba® (-3.7%) vs placebo (-1.7%) at 56 weeks (P<0.001; ITT-LOCF analysis)1
Mysimba® is not indicated for the treatment of diabetes.
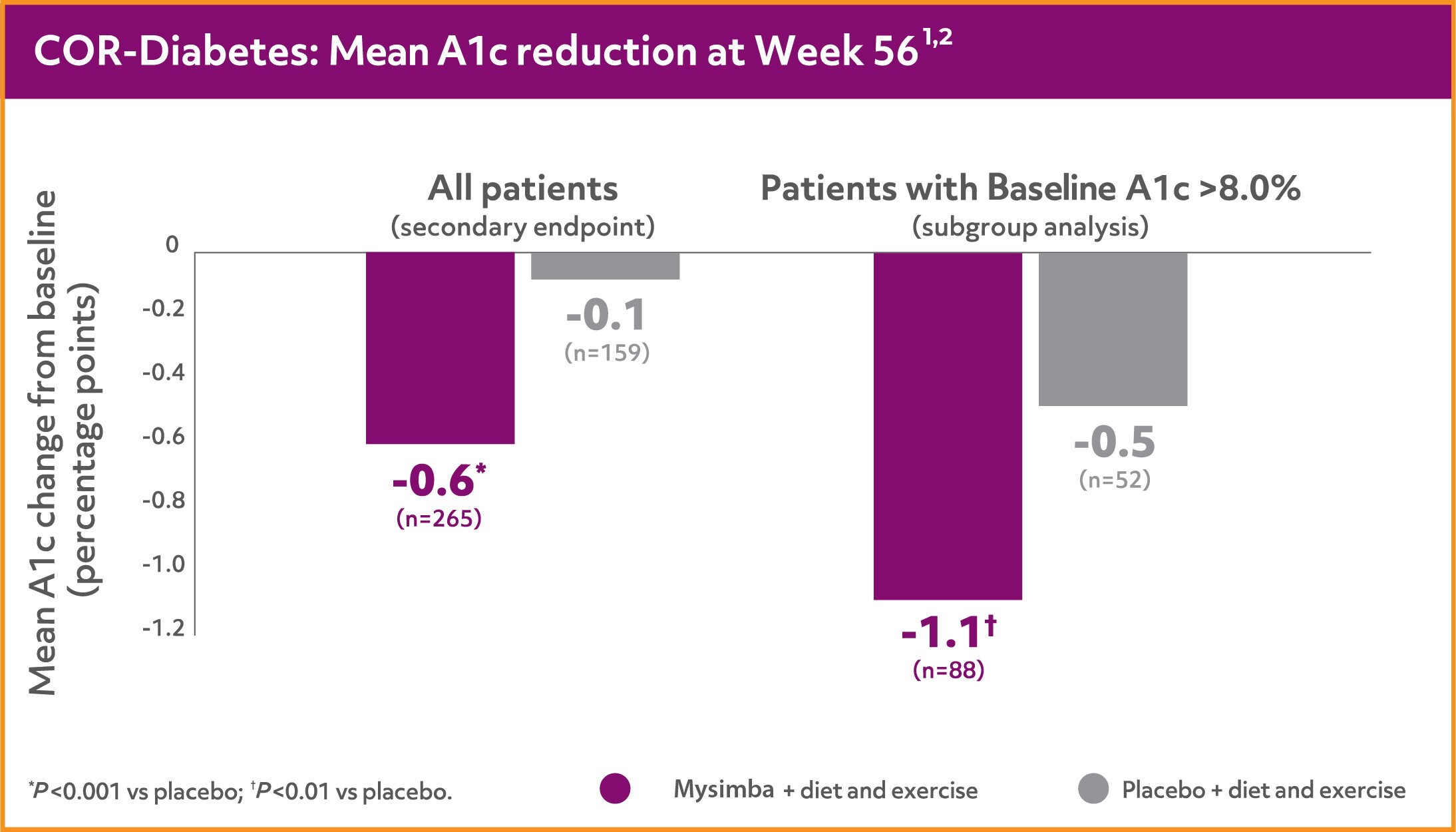
ITT=Intention to treat; mITT: Modified Intention to treat; LOCF= Last observation carried forward; LS=least squares; BOCF; baseline observation carried forward; COR = Contrave Obesity Research; BMOD = Behavioural Modification.
COR-Diabetes was a 56-week, multicentre, double-blind, placebo-controlled study. Overweight or obese patients (BMI ≥27 kg/m2) with type 2 diabetes with or without hypertension and/or dyslipidemia were randomised to Mysimba® 32 mg/360 mg daily or placebo. COR-Diabetes evaluated patients with type 2 diabetes not achieving glycemic goal of an HbA1c <7% either with oral antidiabetic agents or with diet and exercise alone. The co-primary endpoints were percent change from baseline body weight and the proportion of patients achieving ≥5% reduction in body weight at Week 56. LOCF was used for missing data. Mean baseline A1c: Mysimba®: 8.0%; placebo: 8.0%.1
Mysimba® ITT: n=321, placebo ITT: n=166. Average baseline parameters were: Mysimba®: 104.2 kg, 115.6-cm waist circumference; placebo: 105.3 kg, 114.3-cm waist circumference. Patients who completed 56 weeks of treatment: Mysimba®: 52.2%; placebo: 58.8%.1,3
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu .
- Hollander P, Gupta AK, Plodkowski R, et al. Effects of naltrexone sustained- release/bupropion sustained-release combination therapy on body weight and glycemic parameters in overweight and obese patients with type 2 diabetes. Diabetes Care. 2013;36(12):4022–4029.
- Data on file, Currax Pharmaceuticals.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions
Early Responders who had a response at week 16 on average achieved double-digit weight loss at week 561, $
Exploratory analysis results of completers who achieved ≥5% weight loss at week 16 (responders/completers)1, $
Over half (51%) of the patients receiving Mysimba in the modified ITT population lost ≥5% body weight by week 16 vs 19% receiving placebo.1, $
*Average weight loss calculated for responder/completers in the placebo group was not reported individually by study, but rather as a pooled analysis of the placebo groups across COR-I, COR-II, COR-BMOD, and COR-DM.1, $
$ Includes those who had a baseline and Week 56 weight measurement while on study treatment and had lost ≥5% of their baseline body weight by week 16 of treatment
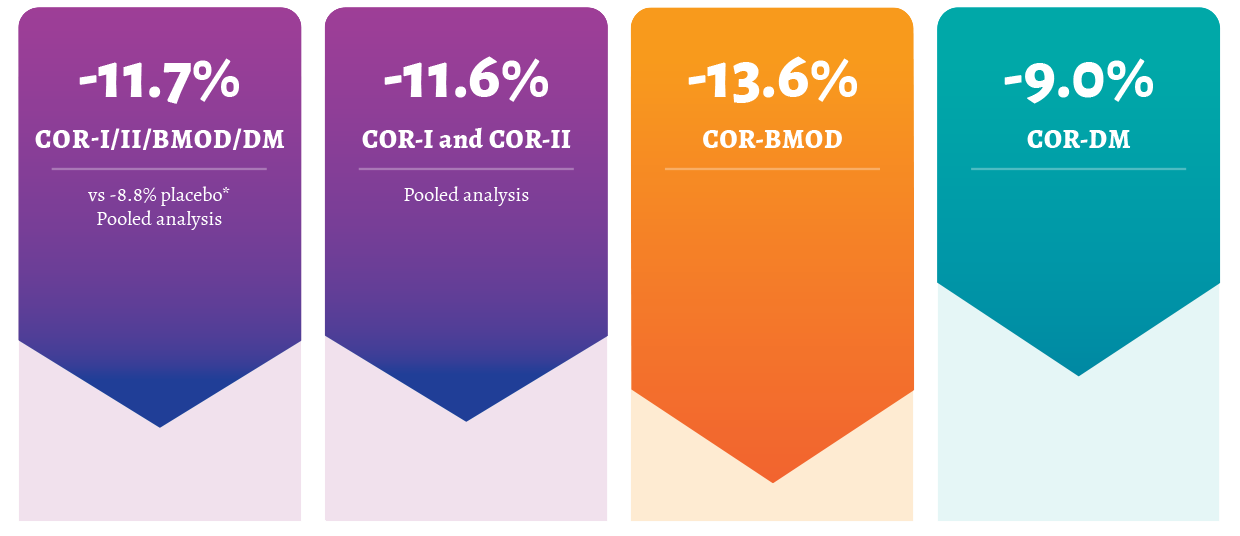
Data analyses: Participant-level responder/completers data from COR-I, COR-II, COR-BMOD, and COR-Diabetes. Exploratory analysis examined the relationship between participant achievement of various weight-loss thresholds at week 8, 12, or 16 of treatment and the associated weight loss at week 56. Efficacy analyses were performed on the modified ITT population (participants with a baseline and at least 1 postbaseline weight measurement while on study treatment with LOCF imputation of missing data; MYSIMBA n=2043, placebo n=1319, for the pooled analysis of the 4 COR clinical trials) and the completers population (participants who had a baseline and week 56 weight measurement while on study treatment; MYSIMBA n=1310, placebo n=763). At week 56, 57% of week 16 responder/completers who received MYSIMBA lost ≥10% body weight vs 39% for placebo.1
COR-II study design: 56-week, multicenter, double-blind, placebo-controlled obesity trial evaluated the effect of MYSIMBA in conjunction with lifestyle modifications in patients randomized to MYSIMBA or placebo. COR-II baseline characteristics were similar to those in COR-I and CORBMOD. The co-primary endpoints were percent change from baseline body weight and the proportion of patients achieving ≥5% reduction in body weight at week 28 and included a blind rerandomization and the addition of a higher dose of naltrexone at week 28 in half of the patients who did not respond adequately to MYSIMBA treatment. Week 56 results (secondary endpoint) presented here include only patients randomized to the approved MYSIMBA dose. 2,3
BMI=body mass index; BMOD=behavior modification; COR=MYSIMBA Obesity Research; ITT=intention-to-treat; LOCF=last observation carried forward; mITT=modified intention-to-treat.
- Fujioka K, Plodkowski R, O’Neil PM, Gilder K, Walsh B, Greenway FL. The relationship between early weight loss and weight loss at 1 year with naltrexone ER/bupropion ER combination therapy. Int J Obes (Lond). 2016;40(9):1369-1375. doi:10.1038/ijo.2016.67
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu.
- Apovian CM, Aronne L, Rubino D, et al. A randomized, phase 3 trial of naltrexone SR/bupropion SR on weight and obesity-related risk factors (COR-II). Obesity (Silver Spring). 2013;21(5):935-943. doi:10.1002/oby.20309
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
IE-MYS-210(1), MYS-0454-001 01/26 DOP: January 2026
The information on this website is intended for Healthcare Professionals who reside in Ireland and Malta only.
Healthcare professionals who reside in Ireland are asked to report any suspected adverse events to: HPRA Pharmacovigilance via www.hpra.ie;
Adverse events should also be reported to Orexigen: +353 1800 849 099 or Currax.Ml@primevigilance.com.
Healthcare professionals who reside in Malta are asked to report any suspected adverse events to: the Malta Medicines Authority via http://medicinesauthority.gov.mt/adrportal or Vivian Corporation Ltd. via pv@viviancorp.com.
Adverse events should also be reported to Orexigen: +356 800 62176 or Currax.Ml@primevigilance.com.