Emotional Eating – a challenge for weight loss success
When it comes treating obesity, one size does not fit all.1 Every patient is different and their treatment should be too. Different causes of obesity create unique challenges for weight-loss success. The Acosta et al observational study found that an individualized treatment approach based on 4 phenotypes was associated with significantly greater weight loss after 12 months compared with the non–pheno type-guided groups.1
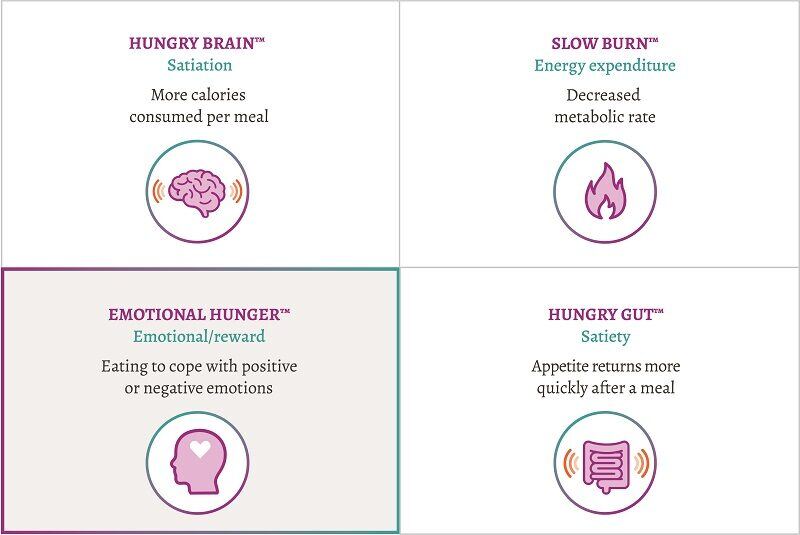
For some patients, this means addressing their cravings that can be associated with emotional eating which occurs when someone eats to cope with positive or negative emotions.2,3 This phenotype is called Emotional Hunger.
Consider Mysimba for your patients with emotional hunger / eating. Mysimba helps control hunger and cravings due to its mode of action in the brain.*
Mysimba is designed to reduce hunger and control cravings so your patients with obesity can lose weight and keep it off.2,4*
- Acosta A, Camilleri M, Dayyeh BA, et al. Selection of antiobesity medications based on phenotypes enhances weight loss: a pragmatic trial in an obesity clinic. Obesity (Silver Spring). 2021;29(4):662-671. doi:10.1002/oby.23120
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu .
- Le Roux et al, Algorithm: Choice of Obesity Pharmacotherapy, Chapter: Pharmacotherapy in Obesity Management, ASOI Clinical Practice Guideline for the Management of Obesity in Adults in Ireland, p15.
- Greenway FL. Physiological adaptations to weight loss and factors favouring weight regain. Int J Obes (Lond). 2015;39(8):1188-1196. doi:10.1038/ijo.2015.59
*The exact neurochemical appetite suppressant effects of naltrexone/bupropion are not fully understood.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions
IE-MYS-216(1), MYS-0454-001 01/26 DOP: January 2026
The information on this website is intended for Healthcare Professionals who reside in Ireland and Malta only.
Healthcare professionals who reside in Ireland are asked to report any suspected adverse events to: HPRA Pharmacovigilance via www.hpra.ie;
Adverse events should also be reported to Orexigen: +353 1800 849 099 or Currax.Ml@primevigilance.com.
Healthcare professionals who reside in Malta are asked to report any suspected adverse events to: the Malta Medicines Authority via http://medicinesauthority.gov.mt/adrportal or Vivian Corporation Ltd. via pv@viviancorp.com.
Adverse events should also be reported to Orexigen: +356 800 62176 or Currax.Ml@primevigilance.com.