Mysimba is specifically formulated to help control hunger and cravings1
Mysimba targets 2 key areas of the brain to help reduce hunger and control cravings, so patients can lose weight and keep it off.1*
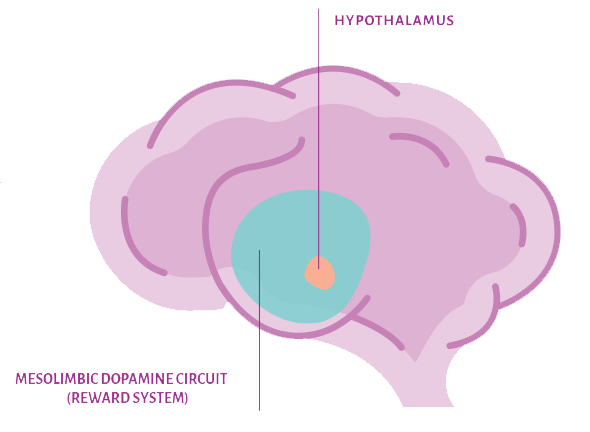
Mysimba is considered to act as a reward system regulator . It contains prolonged-release (PR) naltrexone and bupropion that targets: 1*
- Mesolimbic dopamine circuit (reward system)2,3
Involved with feeling pleasure during rewarding experiences like eating. This may cause intense cravings, making food a source of comfort. - Hypothalamus4,5
Regulates hunger and satiety signals. This part of the brain balances the body’s energy needs and can be influenced by emotional states, potentially affecting eating behavior.
*Other areas of the brain may be involved. The exact neurochemical appetite suppressant effects of naltrexone/bupropion are not fully understood.1
- Mysimba is a unique, proprietary combination of 2 drugs—naltrexone and bupropion—within a prolonged release (PR) tablet clinically proven to help your patients lose weight and keep it off when combined with diet and exercise. 1,6
- The individual medicines of naltrexone and bupropion are not the same dose or formulation used in Mysimba and are not approved for weight loss when used on their own. 1,7,8

Mysimba Mode of Action Video
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information.
Healthcare professionals are asked to report any suspected adverse reactions.
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu .
- Greenway FL. Physiological adaptations to weight loss and factors favouring weight regain. Int J Obes (Lond). 2015;39(8):1188-1196.
- Reichelt AC, Westbrook RF, Morris MJ. Integration of reward signalling and appetite regulating peptide systems in the control of food-cue responses. Br J Pharmacol. 2015;172(22):5225-5238.
- Greenway et al (2010) Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2010 Aug 21;376(9741):595-605.
- Acosta A, Camilleri M, Dayyeh BA, et al. Selection of antiobesity medications based on phenotypes enhances weight loss: a pragmatic trial in an obesity clinic. Obesity (Silver Spring). 2021;29(4):662-671. doi:10.1002/oby.23120
- Wadden TA, Foreyt JP, Foster GD, et al. Weight loss with naltrexone SR/bupropion SR combination therapy as an adjunct to behavior modification: the COR-BMOD trial. Obesity (Silver Spring). 2011(1);19:110-120.
- Bupropion SPC available on HPRA.ie
- Naltrexone SPC available on HPRA.ie
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
IE-MYS-209(1), MYS-0454-001 01/26 DOP:January 2026
The information on this website is intended for Healthcare Professionals who reside in Ireland and Malta only.
Healthcare professionals who reside in Ireland are asked to report any suspected adverse events to: HPRA Pharmacovigilance via www.hpra.ie;
Adverse events should also be reported to Orexigen: +353 1800 849 099 or Currax.Ml@primevigilance.com.
Healthcare professionals who reside in Malta are asked to report any suspected adverse events to: the Malta Medicines Authority via http://medicinesauthority.gov.mt/adrportal or Vivian Corporation Ltd. via pv@viviancorp.com.
Adverse events should also be reported to Orexigen: +356 800 62176 or Currax.Ml@primevigilance.com.

