Mysimba is subject to additional monitoring1 and each patients’ suitability should be assessed with consideration of potential contraindications.
Please refer to the Mysimba Physician Prescribing Checklist on this website or on www.medicines.ie (Ireland) or https://medicinesauthority.gov.mt/rmm (Malta)
- Mysimba 8 mg/90 mg prolonged-release tablets Summary of Product Characteristics available on medicines.ie or www.ema.europa.eu
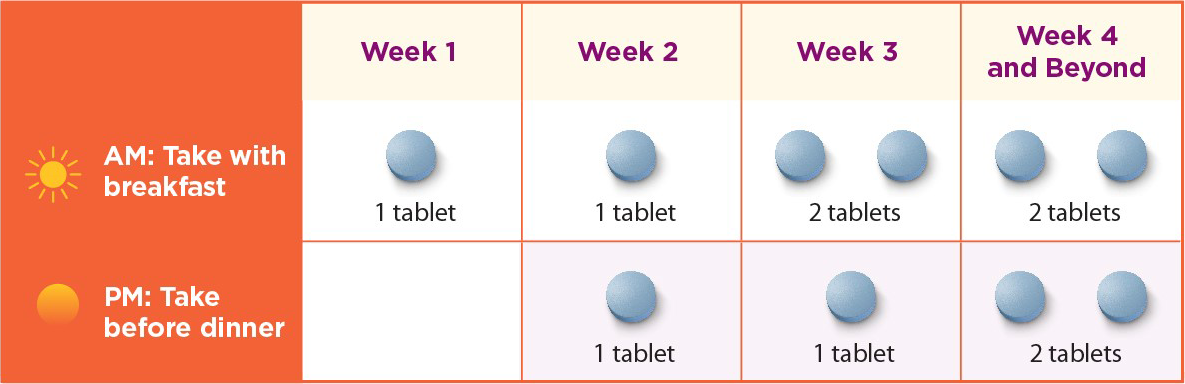
‡Dose adjustments are needed for patients with moderate, severe, or end-stage renal impairment, and hepatic impairment. Maximum daily doses are as follows: Moderate or severe renal impairment: 2 tablets per day (1 in the AM, 1 in the PM). Mild hepatic impairment: 2 tablets per day (1 in the AM, 1 in the PM). End-stage renal disease or severe hepatic impairment: contraindicated. Moderate hepatic impairment: not recommended for use in these patients.
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions
IE-MYS-214(1), MYS-0454-001 01/26 DOP: January 2026
The information on this website is intended for Healthcare Professionals who reside in Ireland and Malta only.
Healthcare professionals who reside in Ireland are asked to report any suspected adverse events to: HPRA Pharmacovigilance via www.hpra.ie;
Adverse events should also be reported to Orexigen: +353 1800 849 099 or Currax.Ml@primevigilance.com.
Healthcare professionals who reside in Malta are asked to report any suspected adverse events to: the Malta Medicines Authority via http://medicinesauthority.gov.mt/adrportal or Vivian Corporation Ltd. via pv@viviancorp.com.
Adverse events should also be reported to Orexigen: +356 800 62176 or Currax.Ml@primevigilance.com.