Introducing Altavita®
Comprehensive care for a broad range of patients – adults, adolescents, children, pregnancy and breastfeeding and those with swallowing difficulties.¹⁻⁵
Click here for further information about patients who are at risk of vitamin D deficiency

Full range of Altavita SmPC’s available on medicines.ie or www.ema.europa.eu
* Doses of 1,000 – 2,000 IU/day may be required to prevent deficiency in some women (see SmPC’s for details of patients at risk of vitamin D Deficiency). Even higher doses may be required during breast-feeding if women choose not to give the infant a vitamin D supplement. †Vitamin D deficiency = 25 (OH) D 25 nmol/l (See SmPC’s)
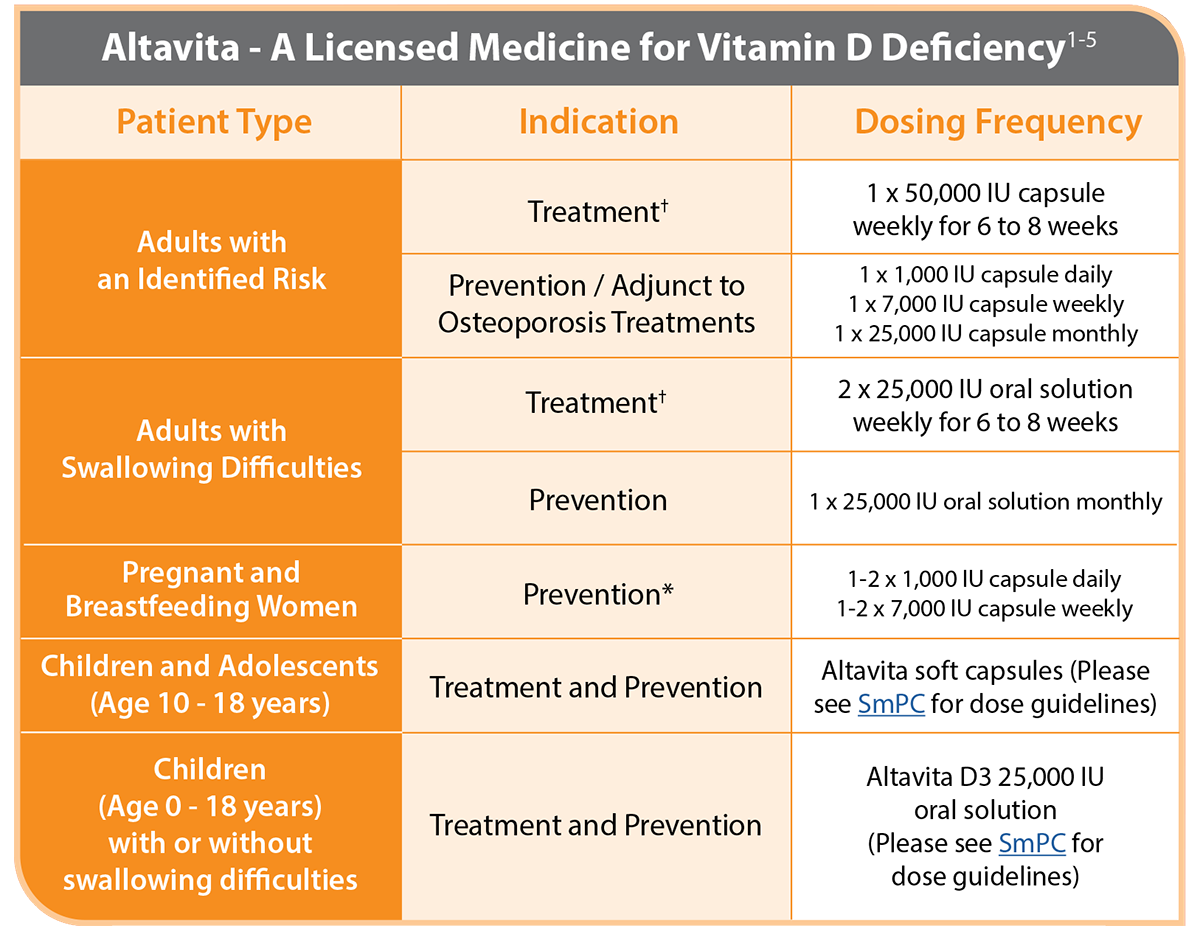
Altavita D3 is indicated for the Prevention and Treatment of Vitamin D Deficiency¹⁻⁵
Prevention of Vitamin D Deficiency
Altavita D3 offers a choice of 1,000 IU daily, 7,000 IU weekly or 25,000 IU monthly vitamin D3 medicines for the Prevention of Vitamin D Deficiency.¹⁻⁴
You can discuss with your patient which of these presentations ties in with their routine. This can aid patient compliance with their medicine.
Treatment of Vitamin D Deficiency
Altavita D3 offers a choice of 1 x Altavita 50,000 IU soft capsules or 2 x Altavita 25,000 IU soft capsules for the treatment of vitamin D deficiency (taken weekly for 6-8 weeks). ³⁻⁵
- Consilient Health Ltd, Altavita® D3 1,000 IU soft capsules, SPC – available on medicines.ie
- Consilient Health Ltd, Altavita® D3 7,000 IU soft capsules, SPC – available on medicines.ie
- Consilient Health Ltd, Altavita® D3 25,000 IU soft capsules, SPC– available on medicines.ie
- Consilient Health Ltd, Altavita®D3 50,000 IU soft capsules, SPC – available on medicines.ie
- Consilient Health Ltd, Altavita® D3 25,000 IU oral solution, SPC – available on medicines.ie
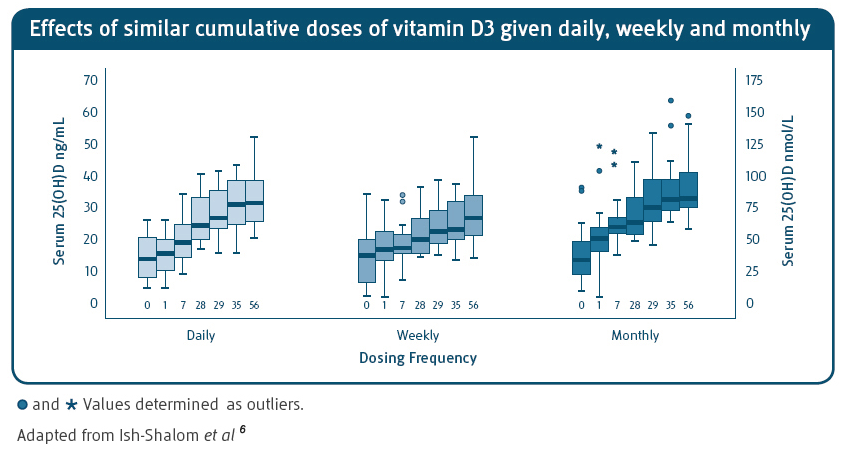
- Ish-Shalom S et al. Comparison of daily, weekly, and monthly vitamin D3 in ethanol dosing protocols for two months in elderly hip fracture patients. J Clin Endocrinol Metab 2008; 93(9): 3430-3435.
IE-ALT-351(3) November 2025
Adverse events should be reported. Reporting forms and information are available from HPRA Pharmacovigilance on the HPRA website www.hpra.ie.
Adverse events should also be reported to Consilient Health at drugsafety@consilienthealth.com or 012057766.
The information on this website is intended for Healthcare Professionals who practice in the Republic of Ireland only.