WHY USE CITIDARON
Why Use Citidaron®?
- The active ingredient of Citidaron® (cytisinicline) has been used as a cessation treatment in Eastern Europe for decades1
- A meta-analysis that included 7 studies of cytisinicline published between 1968 and 2011 showed that cytisinicline was an effective treatment for smoking cessation (risk ratio = 1.59, confidence interval 1.43 to 1.75)2
In a recent Cochrane review, the chances of quitting smoking with cytisinicline are over twice as high, with a success rate of 13%, compared to a 6% chance with control predominantly at 6 months to 12 months.
- Cytisinicline exhibited a low frequency of serious adverse events during clinical trials, suggesting it is generally well-tolerated by patients.
- The high-certainty evidence from the study underlines a well-defined safety profile for cytisinicline, making it a predictable option for smoking cessation.
Please note that some of the studies referenced in the Cochrane report used dosage regimes outside the licensed dosage regime. Cytisinicline’s previously used name is cytisine.

Summary of findings: Lindson meta-analysis, smoking cessation at 6 months 3
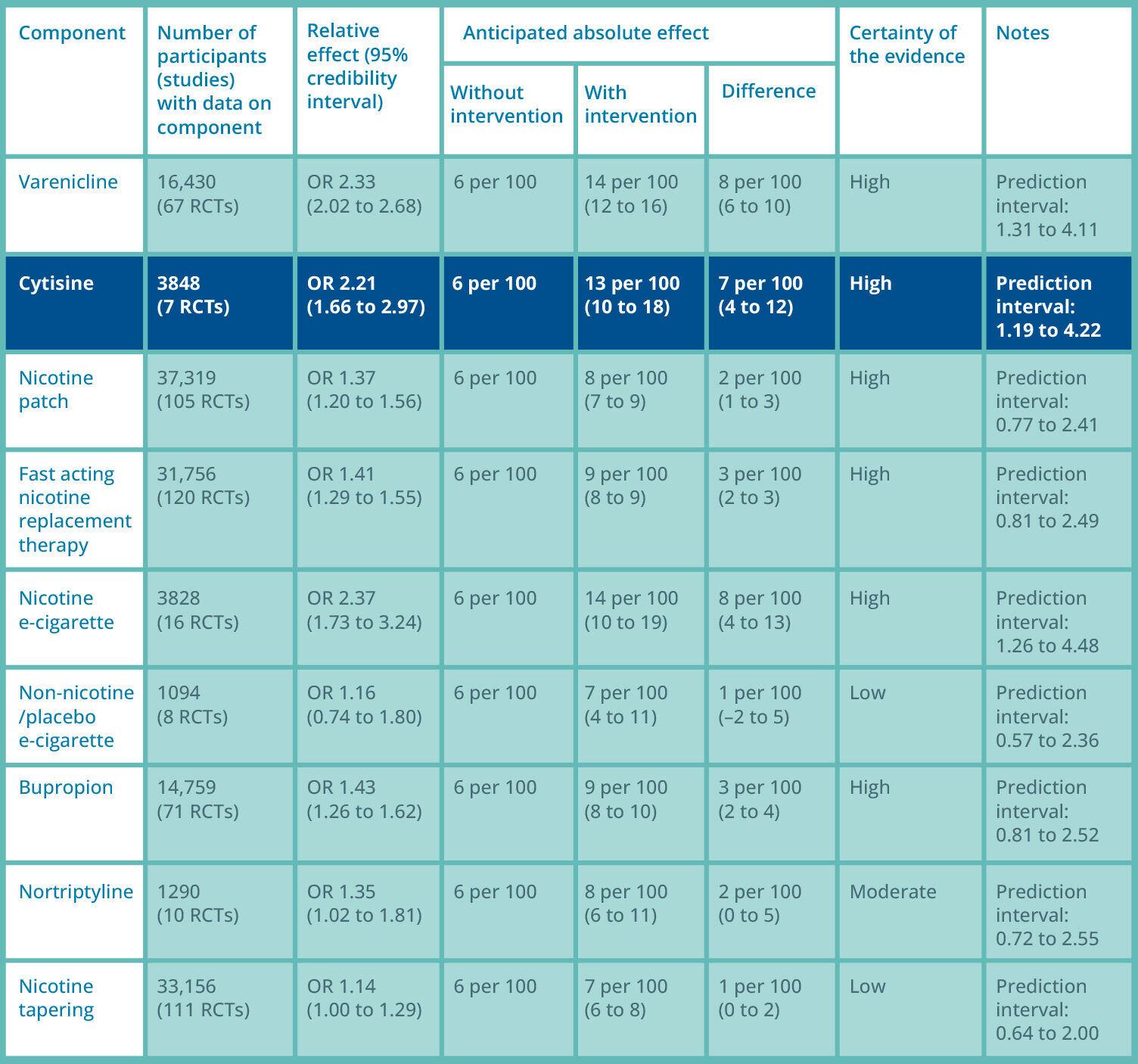
RCT: randomised controlled trial; OR; odds ratio.
Please note some of the studies referenced in the Cochrane report used dosage regimes outside the licensed dosage regime
- Karnieg T, Wang X. CMAJ 2018;190(19):E596.
- Hajek P, McRobbie H, Myers K. Thorax 2013;68:1037-1042.
- Lindson N, Theodoulou A, Ordóñez-Mena JM et al. Cochr Data Syst Rev 2023, Issue 9. Art. No.: CD015226.
Adverse events should be reported. Reporting forms and information are available from HPRA Pharmacovigilance on the HPRA website www.hpra.ie. Adverse events should also be reported to Consilient Health at drugsafety@consilienthealth.com or 012057766.
The information on this website is intended for Health Care Professional who practice in the Republic of Ireland only
IE-CH-1562(2), March 2026
